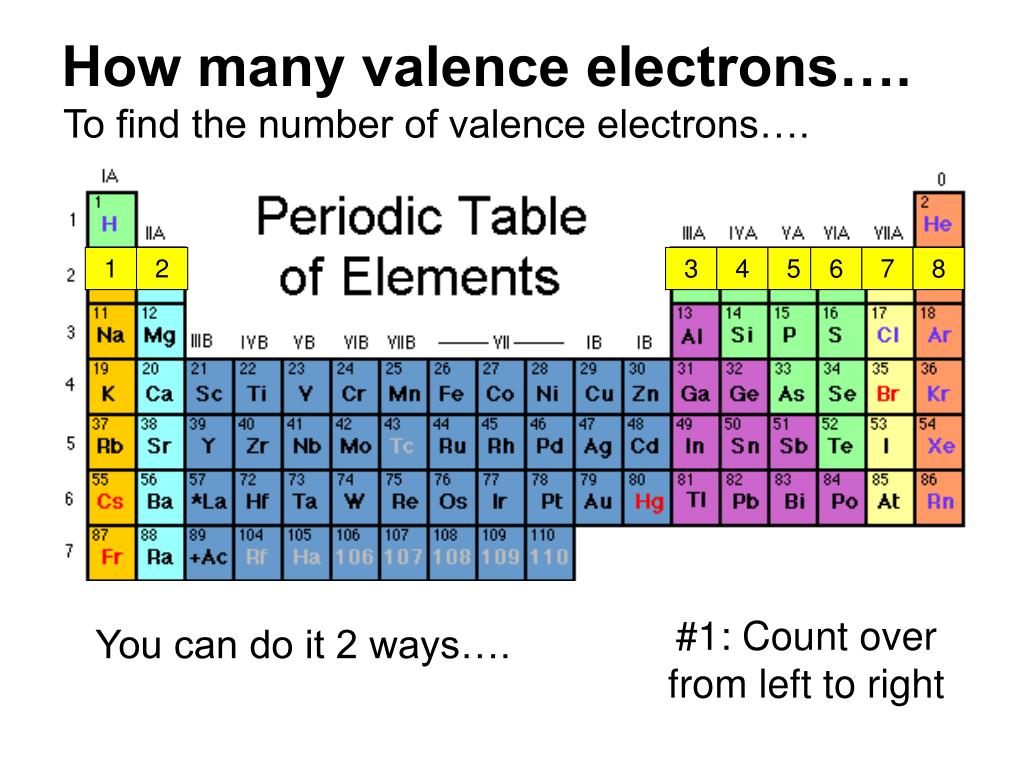
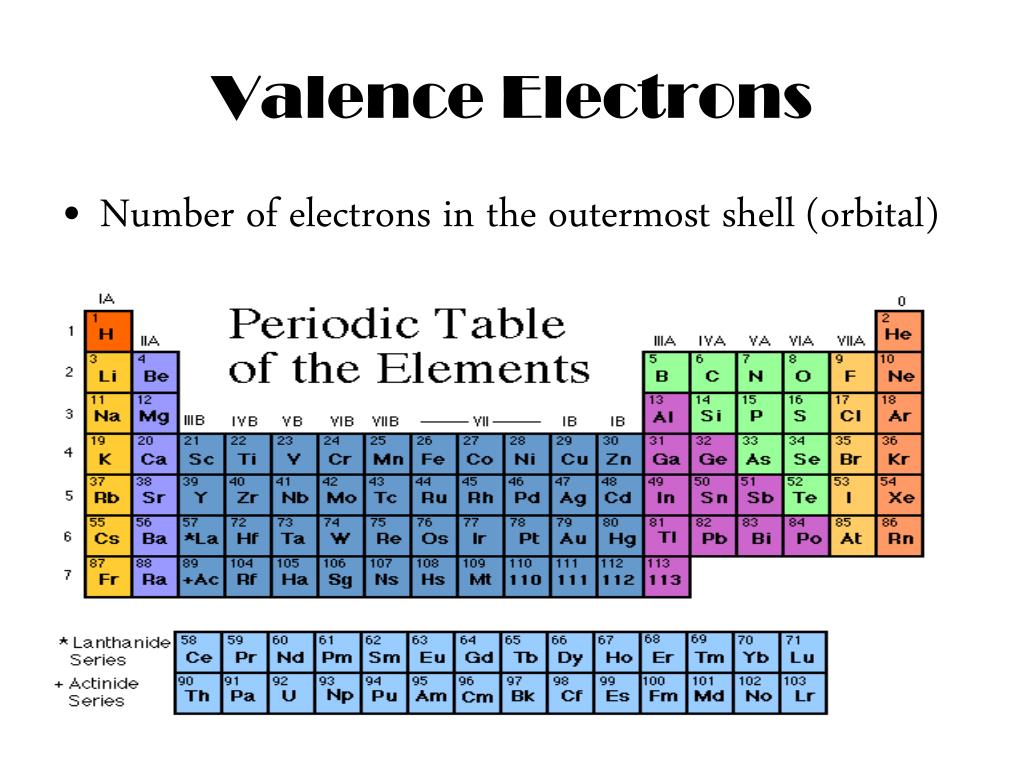
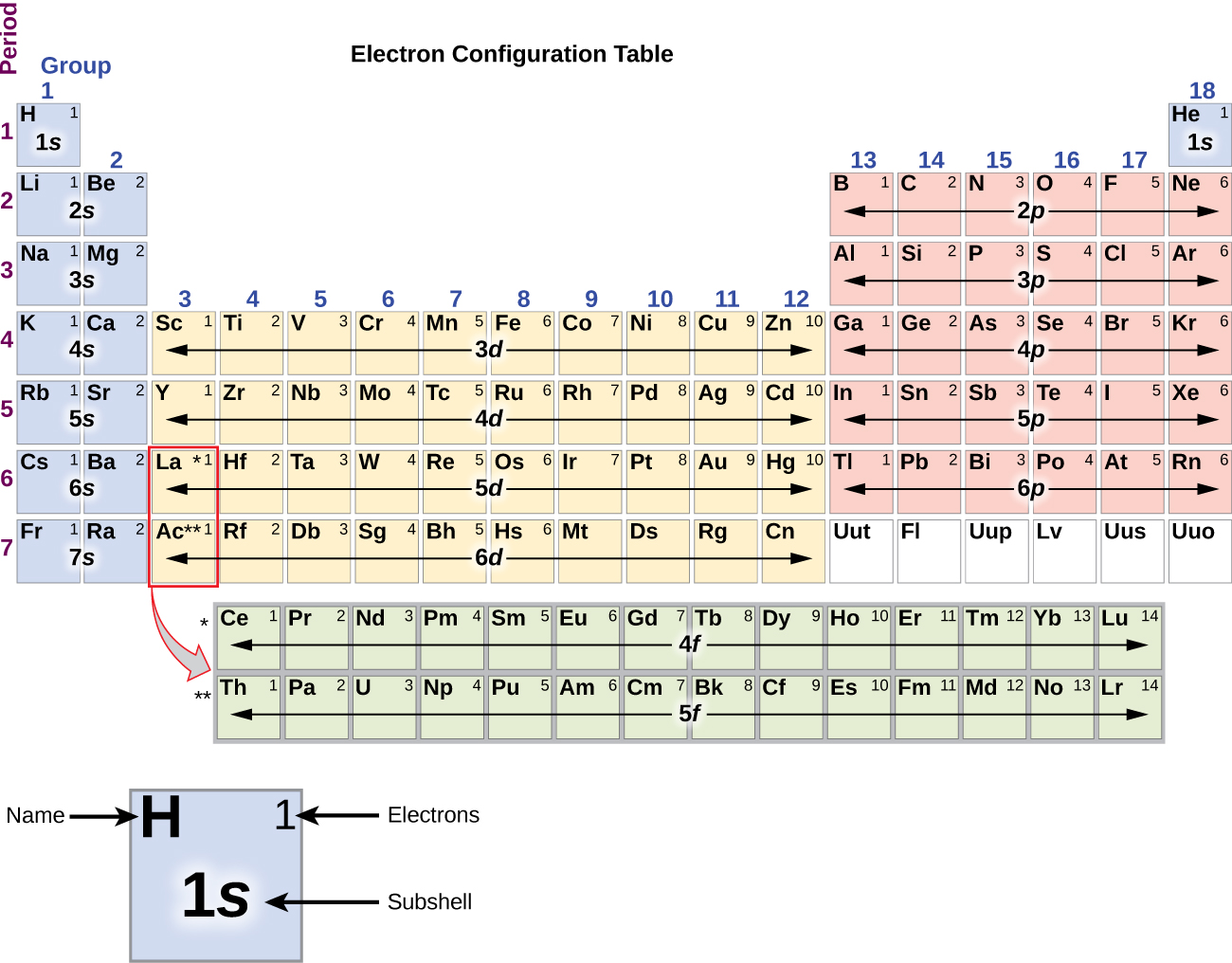
The number of nitrogen valence electrons is 5 Sulfur Valence Electrons So, how many valence electrons does nitrogen have? If we check nitrogen (N) in the periodic table, we will find it in group 15 (Vl) and by applying the fact “The atom’s main group number is equal to the number of valence electrons for these atoms”. Let’s take some examples to apply the periodic table method to find the valence electrons Nitrogen Valence Electrons (Except Helium, which has 2 valence electrons) The following table shows the groups that we can find the number of valence electrons by using the periodic table method and the number of valence electrons for the elements that are located in each group: Periodic Table Group The atomic structure of these elements is different, rigid and need a different way to calculate the number of valence electrons. Note: This method can’t be used with the transition metals which are located in groups number 3,4,5,6,7,8,9,10,11,12. As a result, one atom of sodium has one valence electron. Sodium Valence Electronsįor example, we can find the valency for sodium (Na) which have an atomic number of 11. So, just by checking the number of the group that contains the wanted element, we can know the number of valency for the element which is located in that specific column. Using the periodic table (Valence Electrons – Periodic Table)Īs the periodic table shows lots of important information about the elements, scientists use it to determine the number of valency by checking out the place of elements in the table depending on the facts “All elements in a single vertical column have the same number of valence electrons” and “ T he atom’s main group number is equal to the number of valence electrons for these atoms but ignore the Transition metals which are the elements located in the rectangle-shaped block made by Groups 3 to 12.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
